The neural interface field is no longer dominated by a single use case. Invasive implants for device control and speech restoration still draw much of the public attention, but progress is now spreading across a wider set of interface strategies. These range from subtle limb-based systems designed to restore more natural control of bionic hands, to long-term cortical implants being developed to support stroke rehabilitation.
April has already brought a dense sequence of updates. In the span of just three weeks, CorTec received FDA Breakthrough Device Designation, Phantom Neuro received approval for a first-in-human study, ONWARD Medical raised €40 million, INBRAIN completed enrolment in its first-in-human graphene interface study, and Synchron announced a third clinical trial evaluating its Stentrode. These announcements point to a field advancing across multiple anatomical targets, levels of invasiveness, and clinical pathways.
Phantom Neuro sits slightly outside the narrow BCI category, but inside the same broader neural interface shift. The company is developing a minimally invasive muscle-machine interface designed to restore more natural control of bionic limbs. Instead of placing electrodes in the brain or directly on peripheral nerves, Phantom X uses a small implantable sensor array under the skin in the residual limb to interpret neuromuscular activity and translate it into robotic movement.
In April, Phantom Neuro received approval for its first-in-human Early Feasibility Study in Australia. The CYBORG study will evaluate the safety, efficacy, and usability of Phantom X in up to 10 upper-limb, below-elbow unilateral amputee participants, with procedures taking place at Cabrini Health in Melbourne. The update follows a $19 million Series A round led by Ottobock last year, and positions Phantom as part of a lower-invasiveness interface category moving at full force ahead.
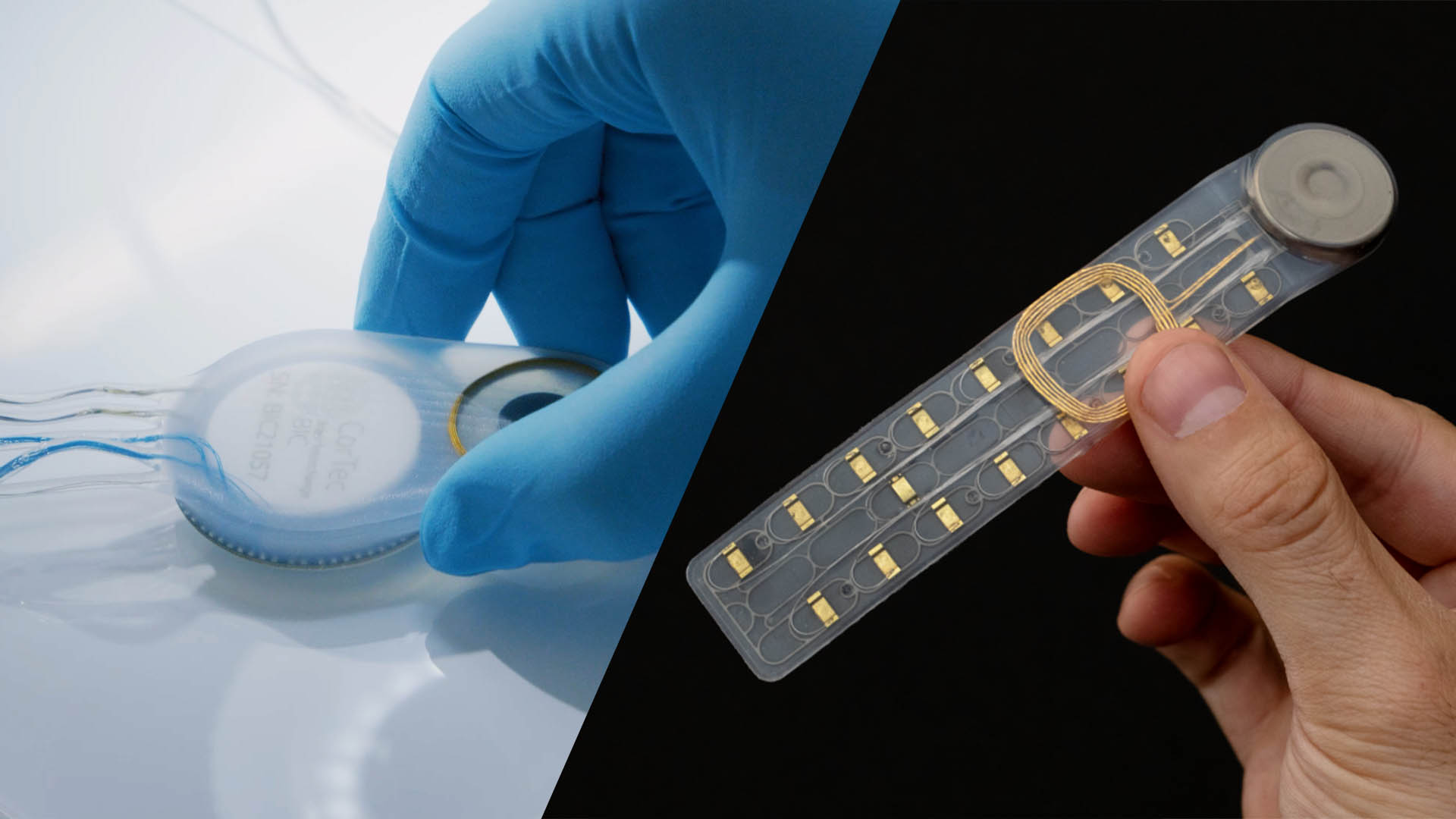
CorTec is advancing a different version of the cortical BCI story. Rather than focusing primarily on communication, cursor control, or direct-to-speech applications, the company is developing Brain Interchange as a fully implantable, wireless, bidirectional interface for therapeutic neurostimulation. The system combines cortical recording with adaptive stimulation.
In April, CorTec received FDA Breakthrough Device Designation for Brain Interchange in stroke motor rehabilitation. The designation covers direct cortical electrical stimulation to support motor recovery in people with chronic stroke-related impairments, and CorTec says it is the first BCI worldwide to receive Breakthrough designation for this indication. The update builds on earlier IDE approval, with the first two patients implanted at Harborview Medical Center in 2025 and 2026, and frames CorTec as part of the therapeutic interface category now moving through early clinical translation.
ONWARD Medical is developing a platform built around spinal cord stimulation, with ARC-EX as a non-invasive system for improving hand strength and sensation after cervical spinal cord injury, and ARC-IM as an investigational implantable system. The company is also developing ARC-BCI, which pairs a brain-computer interface with spinal cord stimulation to restore thought-driven movement.
In April, ONWARD raised €40.6 million through an accelerated bookbuild private placement, including a €25 million investment from EQT Life Sciences. The company said the raise extends its cash runway into Q1 2028, while supporting both ARC-EX commercialization and continued development of ARC-IM. The update makes ONWARD a useful signal for a maturing spinal interface category, where progress is tied not only to clinical studies, but also to sales expansion, regulatory work, and capital discipline.
Synchron remains one of the clearest examples of a minimally invasive BCI strategy. Its Stentrode is implanted through the blood vessels and positioned near the motor cortex, where it records motor intent without requiring open brain surgery. That places Synchron between non-invasive systems and more invasive cortical implants, with the company betting that surgical access and clinical workflow may matter as much as signal density.
In April, Synchron received permission to begin INTENT, its third clinical study evaluating the Stentrode. The study follows SWITCH in Australia and COMMAND in the U.S., where Synchron reported positive one-year results across six implanted patients with severe bilateral upper-limb paralysis. The update also follows the company’s $200 million Series D last year, moving Synchron into a more commercially demanding phase where clinical evidence, reimbursement, and long-term platform integration become central to the BCI pathway.
INBRAIN Neuroelectronics is advancing the materials side of the interface field. The company’s cortical interface is built around ultra-thin graphene electrodes designed to conform to the brain’s surface, capture high-resolution neural signals, and support targeted stimulation. Its differentiation is very much what the interface is made of, with graphene positioned as a way to improve sensitivity, flexibility, density, and signal resolution.
In April, INBRAIN completed enrolment in its first-in-human graphene cortical interface study. Ten patients were recruited, eight were treated surgically, and complete datasets were obtained from all eight treated patients, with no device-related adverse events or device failures reported up to surgical discharge. The update follows the first human use of its graphene interface during brain tumor resection in 2024 and an interim analysis in 2025, which reported no device-related adverse events in the initial cohort and signal acquisition during awake language mapping.