
Europe has an innovation problem. The European Union is increasingly seen as an institutional body that regulates more than it supports innovation. In neurotechnology, that gap is becoming visible. Against the backdrop of China’s first BCI approval, ARIA’s successive multimillion-euro neurotech funding packages, and more than a billion dollars invested in US BCI companies over the last year, Europe’s strategic neurotechnology debate is starting to intensify.
Following two years of research, Brussels-based think tank the Centre for Future Generations (CFG) has released its white paper Towards an EU Neurotechnology Strategy on the 10th of April 2026. The launch coincided with an event co-hosted with the European Brain Commission (EBC), where speakers from across the EU discussed the framework's core pillars: optimizing the EU funding ecosystem, introducing a clearer and more cohesive regulatory framework, and supporting inclusive stakeholder engagement.
The Centre for Future Generations (CFG) is a Brussels-based think tank focused on how Europe governs fast-moving technologies before they are shaped by crisis, market momentum, or external actors. Its work spans AI, biotech, climate interventions, and neurotechnology, with a consistent emphasis on anticipatory governance and public-interest policy. Neurotech has become among the core research areas within that frame.
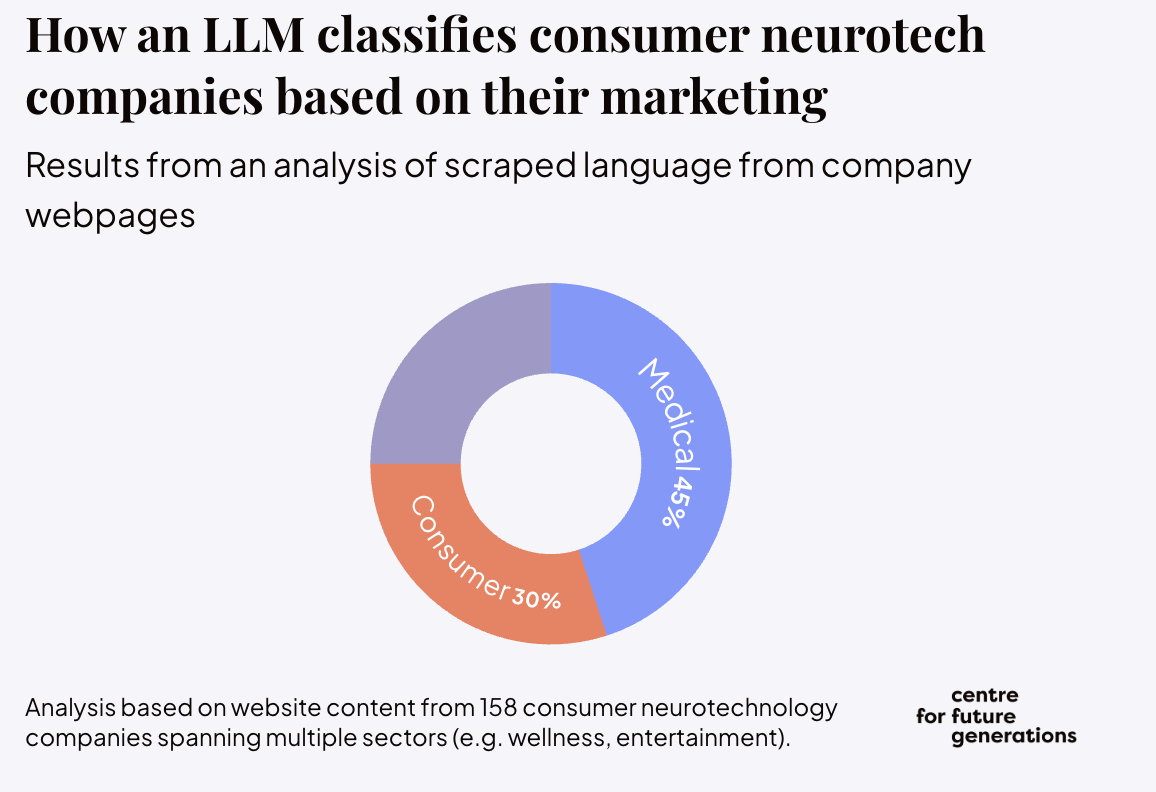
Before the white paper, CFG’s Neurotechnology Programme published a four-part series on the grey zone between wellness and medicine. The series traced how neurotech products move across consumer and clinical categories, and where existing oversight starts to blur. In one part of that work, CFG showed that an LLM classifying consumer neurotechnology based on their marketing language labeled 45% as medical and only 30% as consumer.
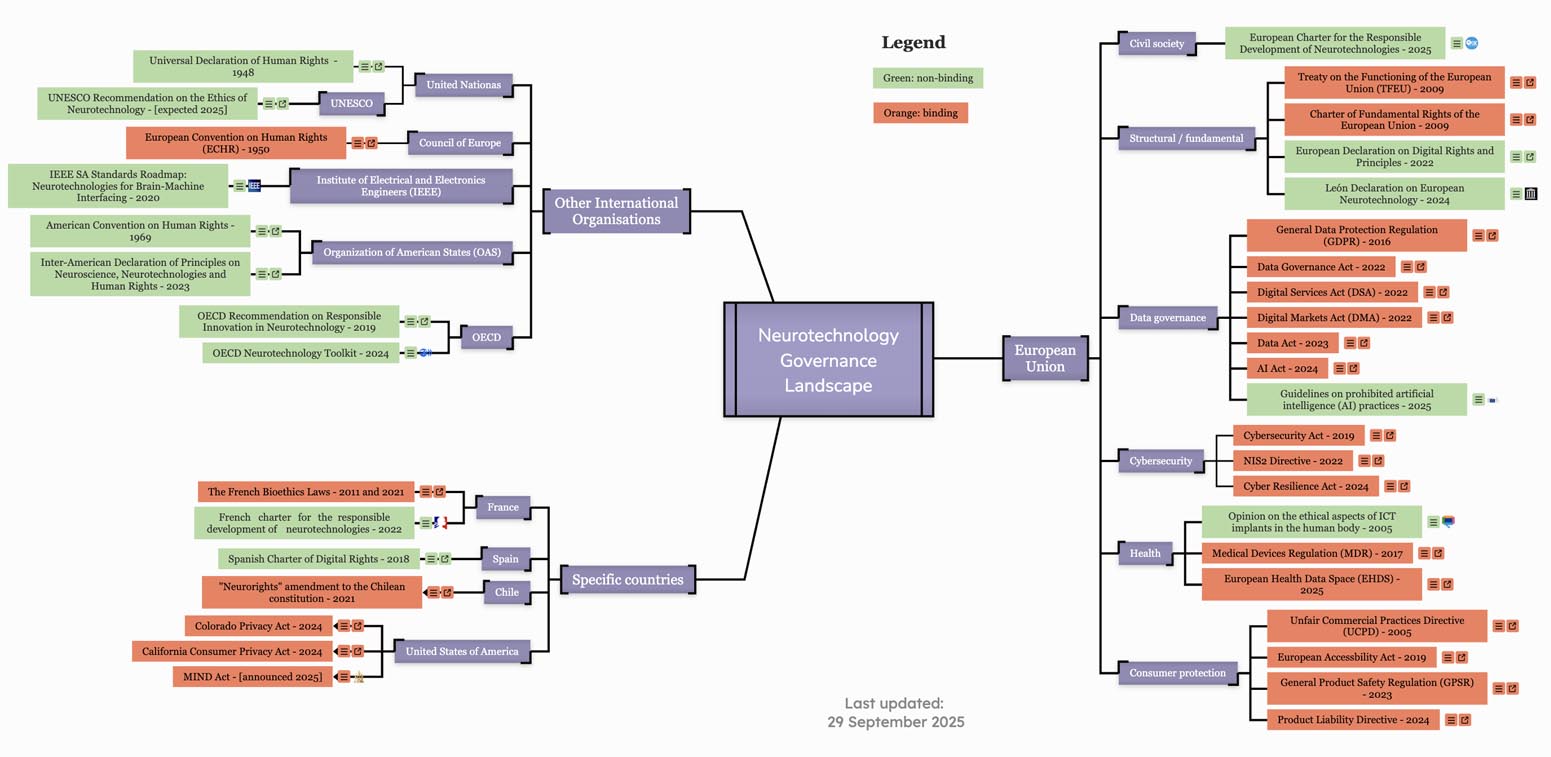
Alongside that, CFG published an interactive governance map that brings together the main EU rules relevant to neurotechnology, from data protection and AI to product safety and medical regulation, while also pointing to selected member-state, international, and non-European frameworks. The map makes visible how fragmented and indirect the current governance landscape still is, with very few neuro-specific rules and most protections relying on broader tech-neutral frameworks.
On this backdrop, the new white paper by Virginia Mahieu, Laura Bernáez Timón, and Paweł Świeboda is presented. It reflects the synthesis of a longer policy effort to make neurotechnology more legible within European institutions and argues that the field now warrants a clearer strategic response at EU level.
The paper is structured around three main pillars: funding, regulation, and public engagement. Across all three, CFG’s argument is that Europe’s problem is not a lack of scientific capacity, but a lack of coordination, continuity, and institutional fit.
On funding, CFG proposes a Neurotechnology Funding Board to coordinate multiphase support across Horizon Europe and public-private partnerships, alongside a pre-competitive Neurotechnology Medicine Platform for shared infrastructure and a “Hub of Hubs” linking Europe’s dispersed centres of excellence.
The problem is framed as structural rather than scientific. Europe accounts for roughly 38% of the global neurotech company base, second only to North America’s 48%, but support rarely follows neurotechnology through its full development cycle. This has resulted in a persistent translational gap between early research and clinical deployment, where many European companies begin to stall.
The regulatory pillar is more legally specific. The paper supports a targeted amendment to Articles 4 and 9 of the General Data Protection Regulation to reduce ambiguity around how neural data is classified, and proposes a distinct legal category for wellness devices outside the Medical Device Regulation. That would give formal shape to a part of the market that currently sits in regulatory limbo, especially as neurotechnology products continue to move across consumer, clinical, and health-adjacent use cases.
The paper pushes the regulatory discussion further than classification alone. It introduces a strategic objective centred on protecting individuals, communities, and vulnerable populations from abuse, manipulation, and misuse, while explicitly defending rights such as mental privacy, cognitive liberty, and informed consent.
On public trust, CFG argues that stakeholder engagement is not separate from governance, but part of it. The paper notes that terms such as “mind-reading,” “brain chips,” and “enhancement” do not merely shape public perception, but also influence policy expectations and institutional responses. From that follows the proposal for a European Neurotechnology Academy, along with a dedicated talent strand within wider scientific programmes, aimed at building a more informed and durable ecosystem around the field.
The grey zone between wellness and medical devices is where the paper develops its most detailed regulatory analysis, and where its proposals would likely have the most immediate commercial implications.
CFG’s market research suggests that consumer-oriented firms now make up between 56% and 60% of the global neurotechnology landscape and have grown more than fourfold since 2010. Many are health-adjacent by design, marketed around sleep, focus, stress, mood, or “brain health” without making formal medical claims, while still leaving room for clinical interpretation by consumers.
The paper is explicit that applying the Medical Device Regulation wholesale to this segment would be disproportionate, and could render large parts of the current market unviable. The MDR hinges on an “intended medical purpose” test that most consumer products do not, and in many cases are not meant to, satisfy.
Instead, the paper proposes a five-part framework: an EU-level oversight mandate; standardized labelling and transparency requirements covering efficacy, safety, data governance, and ethical design; compliance support through sandboxes and model documentation; stricter rules around health-adjacent marketing claims; and interoperability pathways with healthcare systems through the European Health Data Space.
The release of CFG’s white paper, alongside the European Brain Council’s Charter for the Responsible Development of Neurotechnologies, suggests a more coordinated attempt to define Europe’s position in neurotechnology, rather than leaving the field to be shaped primarily through reactive regulation.
For companies operating in the European market, the immediate implications are limited. There is no new legislation on the table yet. Still, the paper maps closely onto debates already underway in Brussels, including the reopening of the Medical Device Regulation and the forthcoming Digital Fairness Act.
In that sense, it clarifies direction; the EU is moving toward a clearer separation between medical and consumer neurotechnology, more explicit treatment of neural data, and greater attention to rights-related risks. For European companies, that points to a more legible regulatory environment. For non-European firms, it sharpens the terms of access.
More broadly, Towards an EU Neurotechnology Strategy is an attempt to push neurotechnology higher on the EU policy agenda and to argue that the field now requires a more dedicated strategic framework. The full paper is available on CFG’s website.