

The brain-computer interface industry is booming. BCI companies brought in just under $2 billion in funding over the past year. Human trials are now running across most of the field’s leading players. Partnerships with AI incumbents, clinical institutions, and platform companies are starting to look like required steps before rollout. It is the stage where the commercial questions become harder to postpone, and where the role of a chief commercial officer starts to define much of future success.

Kurt Haggstrom joined Synchron as Chief Commercial Officer in 2022, becoming part of Tom Oxley’s team building a minimally invasive alternative to the more invasive intracortical systems developed by Neuralink. Synchron has since become one of the defining names in the BCI market. Neurofounders spoke with Haggstrom about the state of the field from the inside, the road to market, the competitive landscape, what public discussion still gets wrong about BCIs, and what the rest of this decade may hold.
How would you describe the phase that Synchron is in today?
I think Synchron, and really the BCI field more broadly, is still in a phase of clinical translation. At this stage, it is not just about understanding what the technology can do, but more importantly, what benefit it can deliver for the patient population it is meant to serve.
Is device control still the main real-world use case Synchron is focused on today, and what aspects of that functionality matter most for patients?
Yes. For people with paralysis, especially upper-limb paralysis, restoring device control is the main goal right now. It is fundamentally about giving people back a degree of independence and autonomy.
Within that, communication remains one of the most important needs. That can mean different things depending on the patient population. For some, especially people who are nonverbal, it is not necessarily about having full conversations. It may be as simple as being able to notify someone, ask for help, or call someone over when needed.
That is why availability matters so much. The system has to be something the individual can activate and use on their own, whenever they want to use it. That sense of autonomy is critical. So it is not just about what the system can do in theory. It is also about whether it is available, usable, and under the patient’s control in the moments that matter.
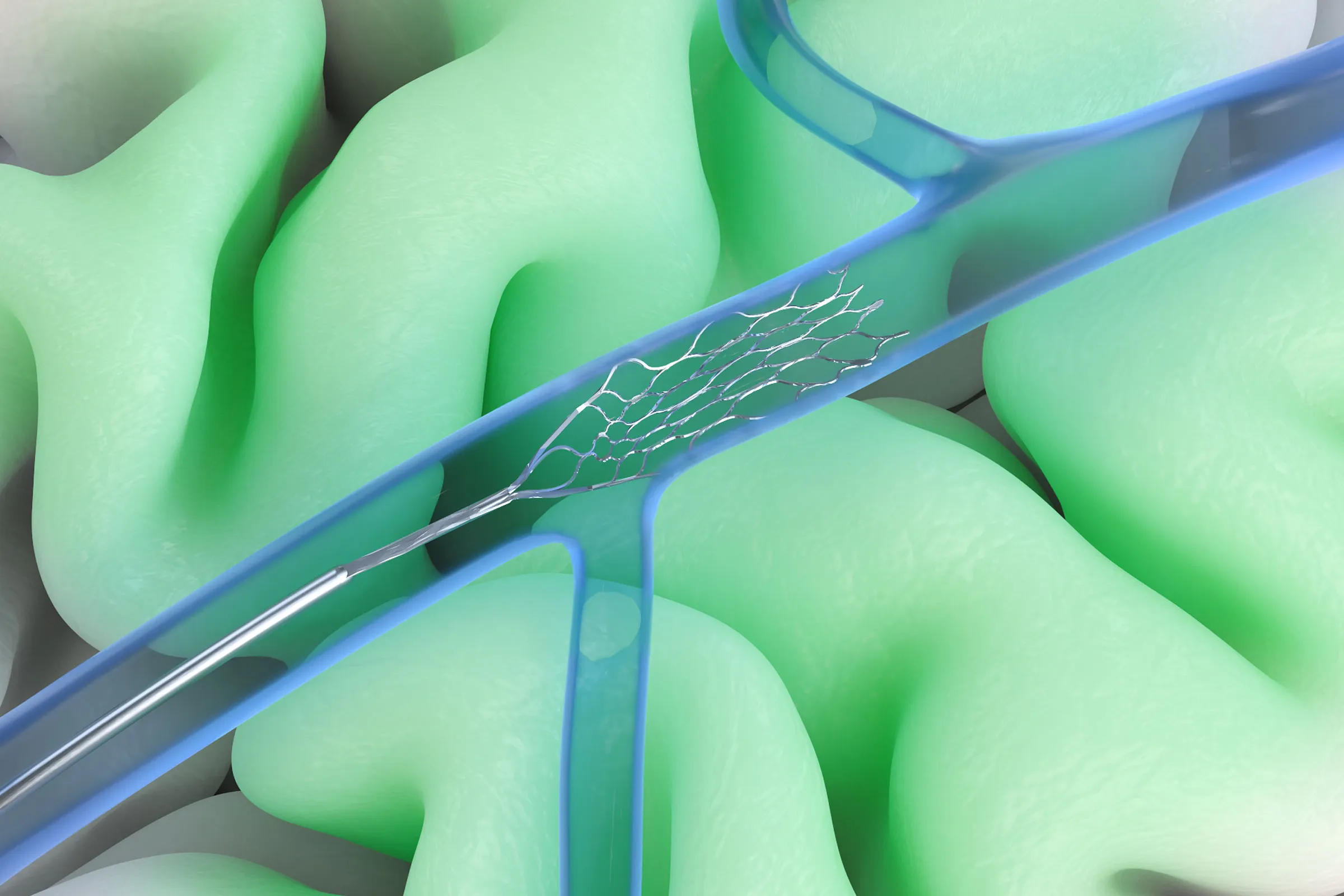
The Stentrode is designed to be minimally invasive, but does it provide enough functionality to cover all of the main use cases?
We sense in a different way than our competitors, and that matters. For example, something like direct-to-speech decoding depends on signals from areas more directly tied to speech production, and that is not where the Stentrode is placed. So that is not really the use case we are targeting.
But if you look at communication more broadly, as well as general computer control and control of robotic systems, then yes, we believe the system can support those kinds of applications.
More broadly, though, those kinds of claims have to be proven in larger clinical trials, and they have to hold up over time. It is not enough to show something on day one or only in the perioperative setting. You need to show stability and utility over months and years.
That is why long-term data matters so much. It is important to evaluate these technologies not just on what they may be able to do acutely, but on what they can sustain over time.
Have there been any unexpected findings from long-term patient use so far?
One of the most striking things has been how much the data science side has continued to evolve in practice.
We have not really seen a limit yet in terms of the kinds of product features that can be built on top of the sensing data. Even after years of use, patients are still discovering capabilities and ways of using the system that they did not have before.
That may look different from one individual to another, because people use their BCI in different ways and a lot of that is learned over time. But to me, one of the most encouraging findings is that we are still learning, still discovering, and have not seen a real plateau. These systems can continue to improve over time.
Do examples like Apple integration make BCIs more understandable and approachable for patients and the broader public?
Yeah. I think bringing big tech, such as Apple, into the discussion early on is really important. Ultimately, those are the platforms and operating systems we are connecting to, so the question is how to make sure the BCI remains a usable control interface as those platforms continue to evolve.
That is why it is important to understand where those companies are going, what accessibility features they are building into their platforms, how we can use those features, and how we think about the future with them. From that perspective, those partnerships are critical.
It has also been encouraging to see how seriously these companies take accessibility. That is true not only of Apple, but of others like Amazon as well. For us, that aligns closely with our own goal of using technology to restore control and independence for people who have lost abilities most of us take for granted.
Are you already thinking about commercialization of the Stentrode?
Yeah, absolutely. When you think about what the business model around a brain-computer interface company will look like, you have to think about the commercial footprint well ahead of time.
A good example is reimbursement. You cannot assume that, once these products reach market, individuals will simply pay for them themselves, especially in systems shaped by Medicare and other payers. So you have to think early about how these technologies are going to be funded, not just in the United States, but in other healthcare systems as well.
That means asking what proof points and what data will be needed not only from a regulatory standpoint, but also from a payer standpoint. It is not enough to show safety and efficacy. You also have to show clinical utility: what does the patient actually gain, how is their health improving over time, and what evidence will be needed to support access and reimbursement? Those are questions you have to think about early, because they shape what data needs to come out of the larger studies.
The other side of it, is the value of the larger models that can be built from neural signals. First, those models can improve the product itself, which should improve performance and adoption over time. But there may also be broader utility there. If you effectively have a front-row seat to a disease such as ALS, for example, can those models generate new insights, digital biomarkers, or a better understanding of disease progression over time?
That is something I find very exciting as well. But it also has to be approached ethically. Patients need to understand what data is being used, what it is being used for, and how it may contribute to different models. Still, I do think there is significant potential there, not just around neural intent, but in signals beyond neural intent as well.
The Precision Neuroscience-Medtronic partnership points toward integrating BCI into existing clinical infrastructure. Is that a path that also interests Synchron?
It is a very different business model.
That approach is less about a long-term brain-computer interface or neuroprosthetic, and more about brain mapping or an intraoperative diagnostic tool. In that context, it is interesting, especially for the kinds of insights it could provide in settings like tumor resection or epilepsy.
It is also a different commercial model, because you are selling into an existing procedure or DRG structure. So it is a shorter-term application of the array, rather than a long-term implant used by an individual over time.
That model may also be useful from a data collection standpoint, not just in improving procedural efficiency or therapeutic outcomes. But it is hard to say one path is simply better than the other. There may be real value in short-term perioperative data collection, but there is also a very different kind of value in a product that stays with the patient continuously over months or years.
So I think you are going to see different paths emerge, with different kinds of utility depending on the model. For us, though, we currently see more value in the long-term implant, and that continues to be our focus.
There are now several companies pursuing long-term implants, each with a very different approach. Do you still see them as direct competitors, or do you think the field will split across different modalities and use cases?
I think there will be room in the market for multiple players. I do not think this is a winner-takes-all space.
That said, I absolutely do view them as competitors. But there are also meaningful differences in approach. For us, the biggest differentiator is that we do not require an open surgical procedure. We take a minimally invasive path, and that matters.
I have spent more than 20 years in medicine watching open procedures shift toward less invasive ones, particularly in fields like cardiology. As treatment moved from open procedures to catheter-based access through the radial or femoral artery, you saw not only safety improve, but also patient willingness to undergo treatment increase.
I do not think this is a winner-takes-all-space
We are already seeing some version of that in the communities we serve. People are more open to a less invasive approach than to a craniotomy or something placed directly into the brain. So I do think that is a meaningful point of differentiation for us.
At the same time, I also think the market will likely split across different applications and competitors. There is enough need, and enough variation in patient and product requirements, that multiple approaches can coexist. And I think it is important for patients to have those options.
There is also a more sensationalist, non-medical narrative around BCIs. How do you view that side of the field, and is it something Synchron is interested in?
At Synchron, we are very patient-centric.
That is not to say there are no possible applications beyond people with medical needs. There probably are. But when I think about the horizon for BCI, and for us at Synchron, it is filled with the stories of people who actually need this technology.
So for me, it is difficult to look too far beyond that toward able-bodied use or so-called superhuman applications. That is not what shapes our day-to-day work. It is not our ethos and it is not our culture at Synchron.
What do you think the public and the media still get wrong about BCIs?
One important point is that this is a medical device category. These are Class III medical devices, which means they fall under the highest level of regulatory scrutiny from bodies like the FDA.
That matters because questions around data privacy, safety, efficacy, and patient benefit are not secondary issues. They are central to the process. These technologies cannot come to market at scale without going through that level of rigor and oversight.
So I think it is important to push back on the idea that this is something that will suddenly appear overnight, with devices being implanted at scale. That is not how this works. These systems require years of study, years of analysis, and a great deal of evidence around safety, benefit, and robustness.
Do you see a future where BCIs become fully non-invasive, or do implanted systems still make more sense for the patients you are targeting?
EEG has been around for a long time. The question is whether it can deliver the fidelity and robustness needed for high-utility applications.
In theory, if strong enough correlations can be built between models derived from more invasive systems and signals collected through something like EEG, then a less invasive path may become possible over time.
But even then, it would not fully solve the issue of independence and autonomy for the individual. You would still need a wearable, something that has to be positioned or put on. In that sense, it would not function as a true prosthetic, where the individual simply has it, controls it, and can use it on their own.
So there is still a strong argument that being somewhat more invasive at the signal level may create a more seamless and autonomous experience for the patient. That said, I would not suggest that a non-invasive future is impossible. It may become more realistic as the data evolves, but we still have to see how that unfolds.
As CCO, what does success look like for Synchron over the next five years?
For me, success means serving the communities we are targeting.
At a practical level, that means proving that this non-surgical BCI approach can work at scale, getting through the clinical trials, and bringing it to market. More fundamentally, it means helping safeguard what we see as a basic human right: freedom of expression.
That is the vision, the mission, and what we are working toward. And I think we can achieve that over the next five years.