April's Snapshot
"A shift in neuromodulation for MDD"
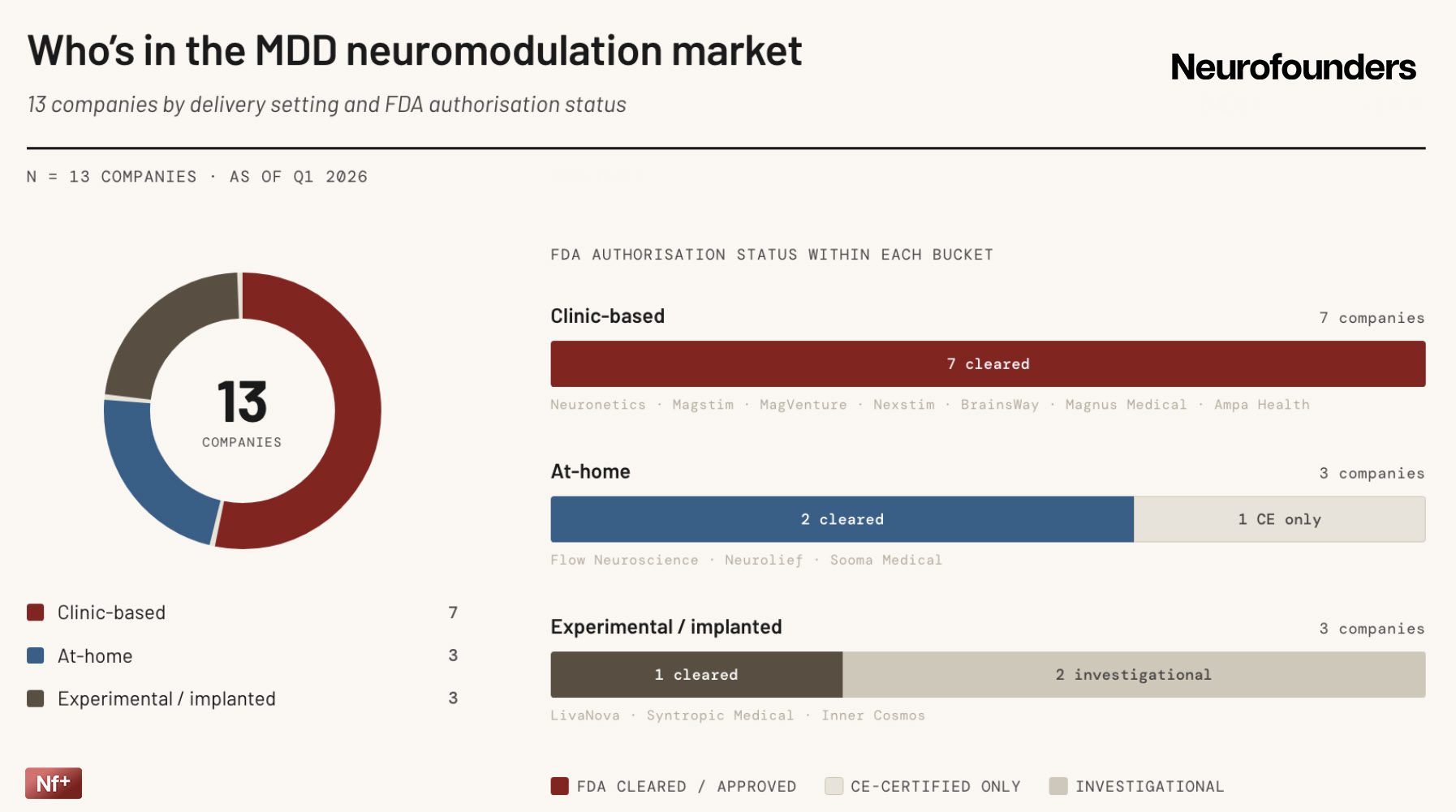
Neuromodulation for MDD is entering a new commercial phase, but the market still rests on an established clinic-based TMS foundation. The main shift is not basic validation anymore, but expansion: especially into adolescent MDD, where several players have recently secured label extensions, making broader deployment a relatively straightforward next step.
At the same time, the category is becoming more layered. Accelerated protocols are making speed and workflow more important competitive factors, while recent FDA approvals for home-use devices suggest that at-home neuromodulation is starting to become a credible part of the market.