

Over the past decades, addiction has slowly changed in the public eye. What was once mostly seen as a matter of bad choices or a failure of character is now understood as a chronic, relapsing disorder rooted deep in neural circuits. Drug exposure recalibrates reward and learning systems in the brain, creating salient relapse cues that are hard to inhibit due to weakened prefrontal control. As a result, the risk of relapse remains present long after withdrawal symptoms disappear.
But while substance use disorder is now much better understood, treatment still relies on low-resolution and often ineffective proxies of addiction. Most clinics operate through self-report, brief screening tools, and urine drug screens. Success is usually measured in binary terms, split between periods of abstinence and relapse. Neurotechnology is starting to change that. With rapid innovation in neuromodulation, neuroimaging, and wearables, a small group of startups is beginning to measure personalized relapse cues, warning of risks in real time and using neuromodulation tools to reduce relapsing behavior.
There is not a single craving centre in the brain. Decades of addiction research have linked reward and motivation processes to dopaminergic signaling, ventral striatal function, and weakened prefrontal systems involved in inhibition, planning, and decision-making. The picture becomes more complex when salience, stress, and memory-related circuitry are added. Yet the most established pattern is one of strong bottom-up drive paired with weakened top-down control.
Breaking through those patterns is much harder than it seems. Addiction-related circuits are activated not just by a substance, but by the people, places, objects, routines, smells, emotional states, and bodily sensations linked to its use. During periods of substance abuse, the hippocampus and amygdala encode a wide range of contextual and emotional associations. These associations resurface repeatedly throughout daily life. And drug-related cues often carry far greater salience than competing environmental stimuli, making them harder to ignore.
The learned cue-substance associations remain active long after acute withdrawal symptoms have disappeared. That makes symptomatic treatment alone not sufficient, as relapsing behaviour can be triggered months or years after substance use has ceased and withdrawal symptoms have disappeared. Meanwhile, it is increasingly recognized that neural and physiological cue responses often do not align with self-reported craving, creating a substantial measurement gap in addiction care.
Substance use disorder has lagged behind adjacent neuropsychiatric categories in terms of neurotechnology, with relatively few tools reaching routine care. The gap goes beyond science. SUD treatment is often delivered in settings shaped by funding constraints, reimbursement gaps, workforce shortages, and regulatory friction. Stigma remains a structural barrier as well, with addiction care frequently separated from mainstream medical settings.
Neurotech innovation is beginning to cluster in two areas. The first is intervention, where rTMS, tDCS, and alternative stimulation paradigms are being studied as ways to modulate craving, executive control, and reward-related circuitry. DBS is also explored, but due to its invasive nature, application is limited to more severe and treatment-resistant cases.
Second is measurement. New neurotech platforms increasingly focus on uncovering biomarkers derived from sleep, physiology, behavior, and proxy signals. Within that group, EEG and event-related potential (ERP) approaches stand out as established techniques for capturing neural cue reactivity more objectively than self-report or behavioral proxies. ERPs isolate the brain’s time-locked response to stimuli, making them useful for tracking how substance-related cues are processed in the first moments after they appear.
But most neurotech work remains early. SUD literature is methodologically heterogeneous, with wide variation in stimulation targets, dosing, task design, substance category, and outcome definitions. Real-world deployment remains limited relative to the enormous scale of disease burden, in part because implementation has to fit fragmented care pathways and inconsistent coverage environments.
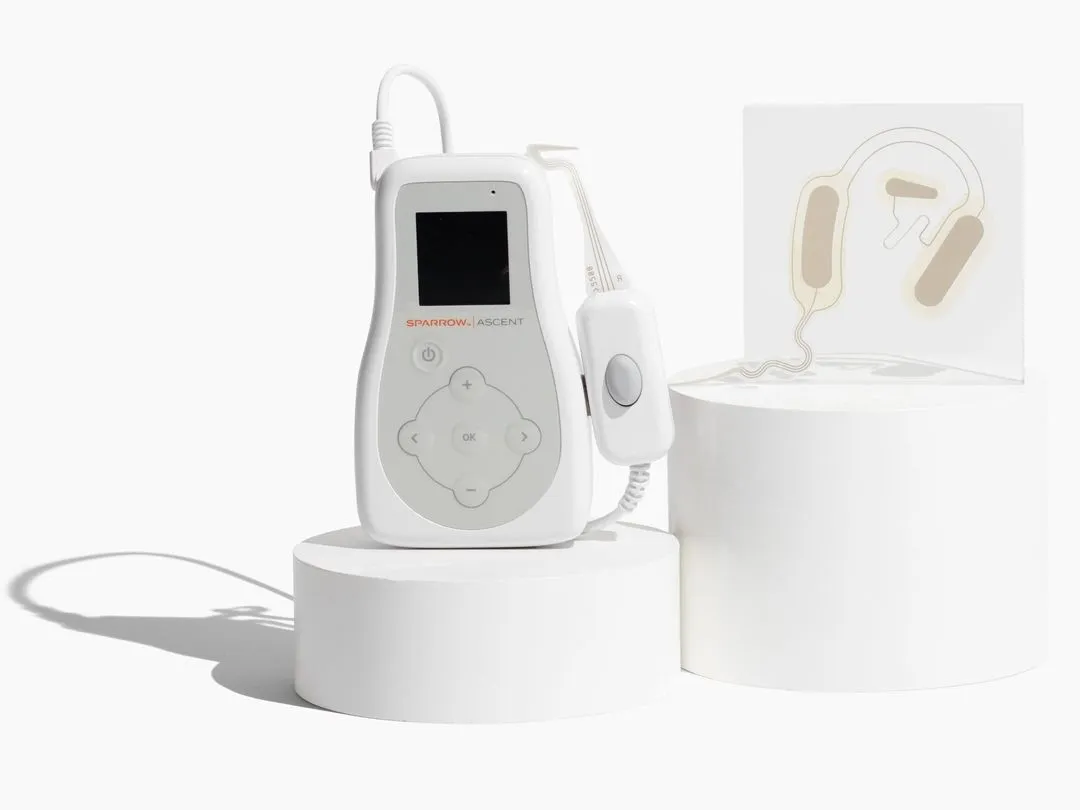
A pragmatic entry point into addiction care is tackling the withdrawal window. For many patients, the first phase of recovery is one of the hardest parts of treatment. Symptom burden can quickly undermine adherence and push recovery off course. In that narrow but important window, Spark Biomedical is building its position. The Dallas-based bioelectronic medicine company developed Sparrow Ascent, a prescription wearable designed to reduce opioid withdrawal symptoms under trained clinical supervision.
Sparrow Ascent uses transcutaneous auricular neurostimulation, delivering electrical stimulation on and around the outer ear. Spark says this approach targets branches of the vagus and trigeminal nerves, with the aim of modulating autonomic and central pathways involved in withdrawal. The product is not positioned as a long-term treatment for opioid use disorder, but as a drug-free adjunct for acute withdrawal symptoms that can be deployed across inpatient, outpatient, and at-home settings.
Within that narrow commercial focus, Sparrow is picking up early clinical and operational traction. Clinical research using Sparrow shows the device reducing opioid withdrawal symptoms by 46% after 60 minutes in a randomized controlled detox setting. The second-generation Sparrow Ascent received FDA clearance in 2023, and by 2024 Spark said it had reached around 50 treatment centers across the U.S. From there, the company has begun extending the same neuromodulation platform into broader investigational settings, including alcohol withdrawal, relapse prevention, and addiction-related comorbidities such as PTSD.
On the measurement front, a clear attempt to turn cue reactivity into a usable clinical tool comes from Dr. Scott Burwell at the University of Minnesota’s Department of Psychiatry and Behavioral Sciences. Much of his work has focused on how the brain responds to personalized cues tied to past substance use, including places, paraphernalia, people, and contexts that slowly accumulate meaning over years of use.
Rather than asking only what someone says they crave, his studies measure ERPs using EEG. This gives Burwell an objective insight into what the brain is doing in the first few hundred milliseconds after cues appear. In addiction research, those signals can reveal a kind of hidden craving response: neural reactivity that remains strong even when someone reports feeling stable or confident in recovery.

In 2019, Burwell founded Neurotype to bring his cue-reactivity work closer to real-world care. The company’s NeuromarkR assessment tool adapts lab-style ERP tasks into a short EEG session in which people with opioid use disorder view sets of neutral and substance-related images while their brain responses are recorded. He aims to help clinicians see which cues most strongly activate a person’s relapse circuitry and how that pattern shifts over weeks or months to personalize treatment.
One theme Burwell emphasized in our conversation is how deeply personalized cues can become. “A colleague recently mentioned how people often have their own paraphernalia for smoking or injecting,” he said. “If it’s your personal device versus a generic pipe, your brain responds much more strongly to the one you’re familiar with.” That degree of individual specificity is part of what makes translating biomarker research into a consistent clinical tool so difficult. But it is also where the value may lie.
Neurotype shows how decades of addiction research might eventually give clinicians a more objective lens on relapse risk, rather than leaving them to rely mainly on interviews and self-report. And with efforts like these, neurotechnology is starting to support a growing population living with deeply embedded patterns of addiction.