
Neurodegenerative therapies have traditionally focused on targeting toxic protein accumulation with molecules. Yet growing evidence suggests that the earliest dysfunction in Alzheimer’s disease arises from disrupted brain network activity, particularly breakdowns in gamma oscillations. These fast brain rhythms synchronize communication across neural circuits and play a critical role in memory, attention, and synaptic plasticity, fuelling growing interest in therapies that restore gamma synchrony.
Last week, Massachusetts-based neuromodulation company Cognito Therapeutics announced an oversubscribed $105 million Series C financing for the development of Spectris, an at-home, non-invasive headset designed to promote gamma oscillations in patients with Alzheimer’s disease. The funding will support advancement of Spectris through its Phase 3 clinical trial, preparation for an FDA submission, and expansion of the platform into additional neurodegenerative indications.
The financing follows clinical studies which demonstrated Spectris’ ability to slow neurodegeneration. In the company’s OVERTURE Phase 2 study, 33 patients with mild-moderate Alzheimer’s disease received visual and auditory 40 Hz stimulation via the Spectris headset. 40 Hz is the frequency that promotes gamma-frequency activity in neural populations. The study met its primary objectives of safety and tolerability, with no serious device-related adverse events reported.
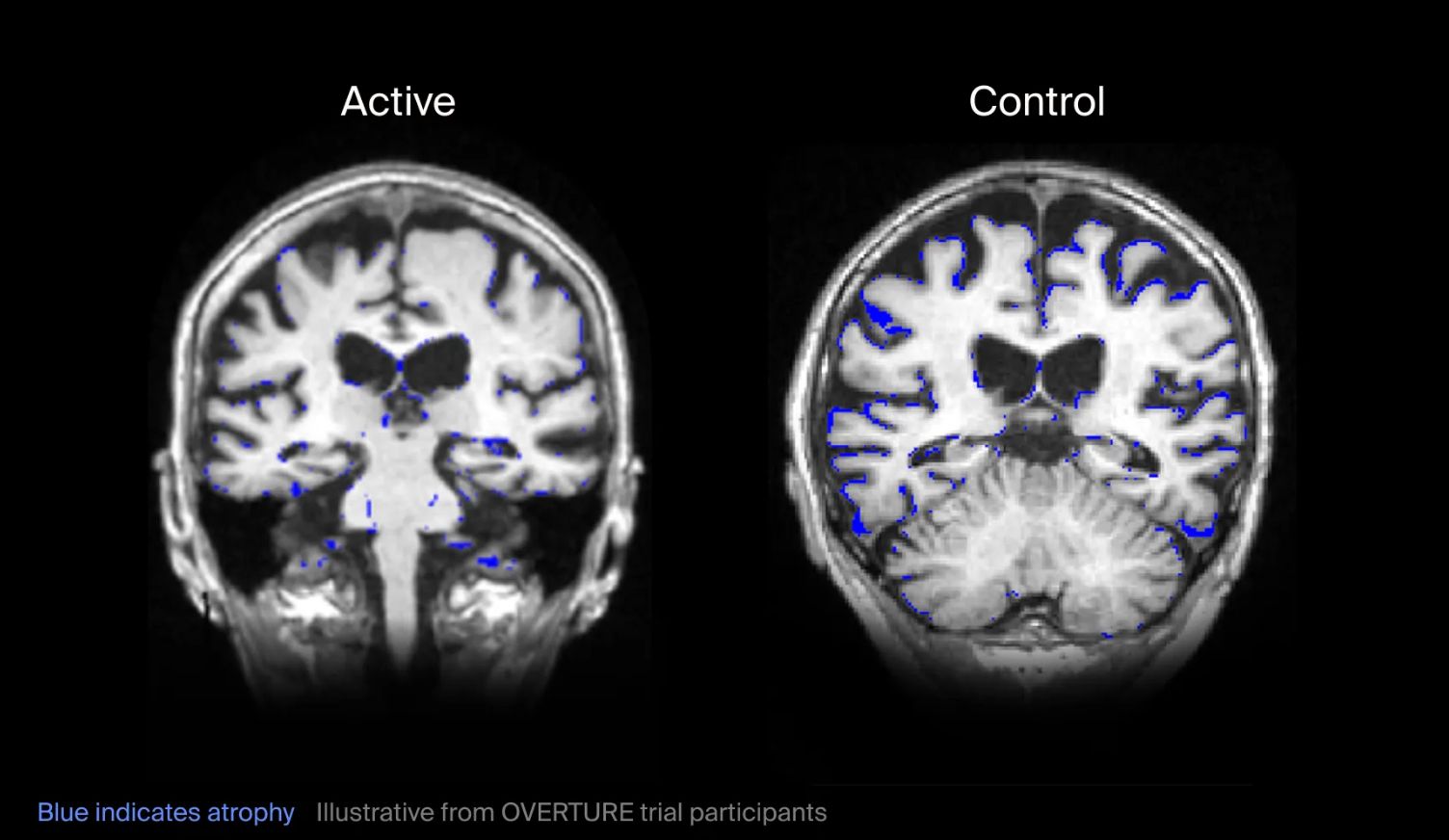
While the trial was not statistically powered to demonstrate clinical efficacy, exploratory analyses showed encouraging signals. Patients in the treatment group (n=33) demonstrated preservation of brain volume on MRI compared with the control group (n=20); a notable finding given that brain atrophy is closely associated with functional decline in Alzheimer's disease and typically progresses faster than in healthy aging. Additional analyses also suggested preservation of white-matter and myelin integrity, along with slower decline in cognition and daily functioning.
Cognito is now advancing its pivotal Phase 3 HOPE trial, which is evaluating the Spectris system in approximately 670 patients with mild-to-moderate Alzheimer’s disease. The therapy has received FDA Breakthrough Device designation, a program intended to accelerate regulatory review for technologies addressing serious conditions with limited treatment options. This designation is expected to streamline the path toward authorization if clinical benefit is demonstrated.
Key questions remain regarding Spectris’s ability to demonstrate robust clinical benefit in larger trials, particularly showing improvements in cognition and daily function rather than biomarkers alone. Device adherence also remains an open question. Cognito reports that patients have completed more than 100,000 treatment sessions with strong compliance, however adherence within structured clinical trial environments does not necessarily translate to long-term use in routine care.
Gamma oscillations have long been recognized as an important marker of brain health. In Alzheimer’s disease, reductions in gamma oscillations have been shown to correlate with cognitive decline and memory impairment. Pre-clinical studies have shown that restored gamma activity promotes neuroprotective effects in animal models, including activation of microglia, the brain’s immune cells, to help clear amyloid aggregates, the hallmark protein deposits of Alzheimer’s disease.
Additional studies have suggested that gamma entrainment may influence long-term potentiation (LTP), a core mechanism of synaptic plasticity that underlies learning and memory and is known to be impaired early in Alzheimer’s disease. Researchers have also observed changes in gene expression associated with synapse formation, neuronal protection, and inflammatory regulation, suggesting that restoring gamma activity may help stabilize neural circuits vulnerable to degeneration.
The Spectris headset restores gamma oscillations by delivering synchronized light flicker and auditory pulses at 40 Hz. This frequency propagates throughout broader neural networks and promotes gamma-frequency activity in neural populations via a phenomenon known as the steady-state sensory response, whereby neurons in the relevant sensory cortex begin firing in synchrony with the rhythm of the stimulus.

Cognito is emerging alongside a small group of companies exploring neuromodulation approaches for Alzheimer’s disease. NeuroEM Therapeutics is developing wearable electromagnetic stimulation devices designed to break up toxic amyloid and tau aggregates, while Sinaptica Therapeutics is advancing personalized transcranial magnetic stimulation to restore disrupted brain networks and enhance neuroplasticity. As these therapies target early stages of disease, their potential impact will depend in part on the ability to diagnose Alzheimer’s earlier, enabled by improved diagnostic tools and more widespread screening.