
The brain-computer interface race is increasingly moving towards clinical translation. That shift brings a harder question into focus: can neural implants maintain stable, high-quality contact with brain tissue over longer periods of time? Harvard-spinout Axoft sits in a cohort of companies trying to answer that question. It is building its interface platform around Fleuron, a bio-inspired material designed to make implants softer and more compatible with neural tissue.
Last week, Axoft announced a $55 million Series A to advance its BCI platform. The funding will support global clinical trial expansion, U.S. regulatory work, and a new facility for mass production of its devices and materials. The raise follows FDA Breakthrough Device Designation, first-in-human studies, clinical collaborations, and early human recordings. $55 million goes into the books as another sizeable BCI round. Yet more importantly, it points to a growing role of materials design in implantable neurotechnology.
Last week, Axoft announced a $55 million Series A to advance its bio-inspired implantable brain-computer interface. The oversubscribed round was led by C.P. Group Innovation, with participation from Alumni Ventures, Stanford President’s Venture Fund, Hillhouse Investment, and Gaorong Ventures. The raise brings Axoft’s total funding to more than $60 million and will be used to expand clinical trials, progress U.S. regulatory approval, and build a facility for mass production of its BCI platform and proprietary materials.
At the center of the platform is Fleuron, Axoft’s proprietary material for creating a softer interface between electronics and neural tissue. The platform is positioned around higher-quality neural data capture, improved biocompatibility, and clinical applications in prognosis and communication for disorders of consciousness. Axoft is also trying to make Fleuron available beyond its own implantable BCI. CEO Paul Le Floch said several industrial and academic groups are already using the material for research and development, adding that Axoft aims to make its “novel material platform widely available.”
That broader materials strategy has been built alongside the company’s device work. Axoft launched Fleuron through an exclusive Stanford licensing agreement, later secured a joint development agreement with Kayaku Advanced Materials, and introduced a microfluidics formulation for biomedical engineering applications including biohybrid devices, organ-on-chip systems, and microfluidics. The company has also opened a subsidiary in Grenoble, adding a European base to its materials and clinical translation efforts.
Axoft was founded in 2021 by Paul Le Floch, Tianyang Ye, and Jia Liu, emerging from Harvard research on ultra-flexible nanoelectronics designed to mimic the mechanical and structural properties of the brain. In 2022, the company launched with $8 million in seed funding and announced FDA Breakthrough Device Designation.
Since then, Axoft has reported first-in-human progress, including the first four cases in its FINESSE study at The Panama Clinic, where its probes were implanted in patients undergoing brain tumor resection and used to record 20 minutes of activity across cortical and subcortical regions.
Axoft’s BCIs have now been tested temporarily in 11 patients worldwide, including four in Shanghai, according to Bloomberg. The company plans to study the device in more patients in China this year and is preparing multiple clinical trials in the country. The data so far remains based on temporary implantation rather than chronic, everyday use.
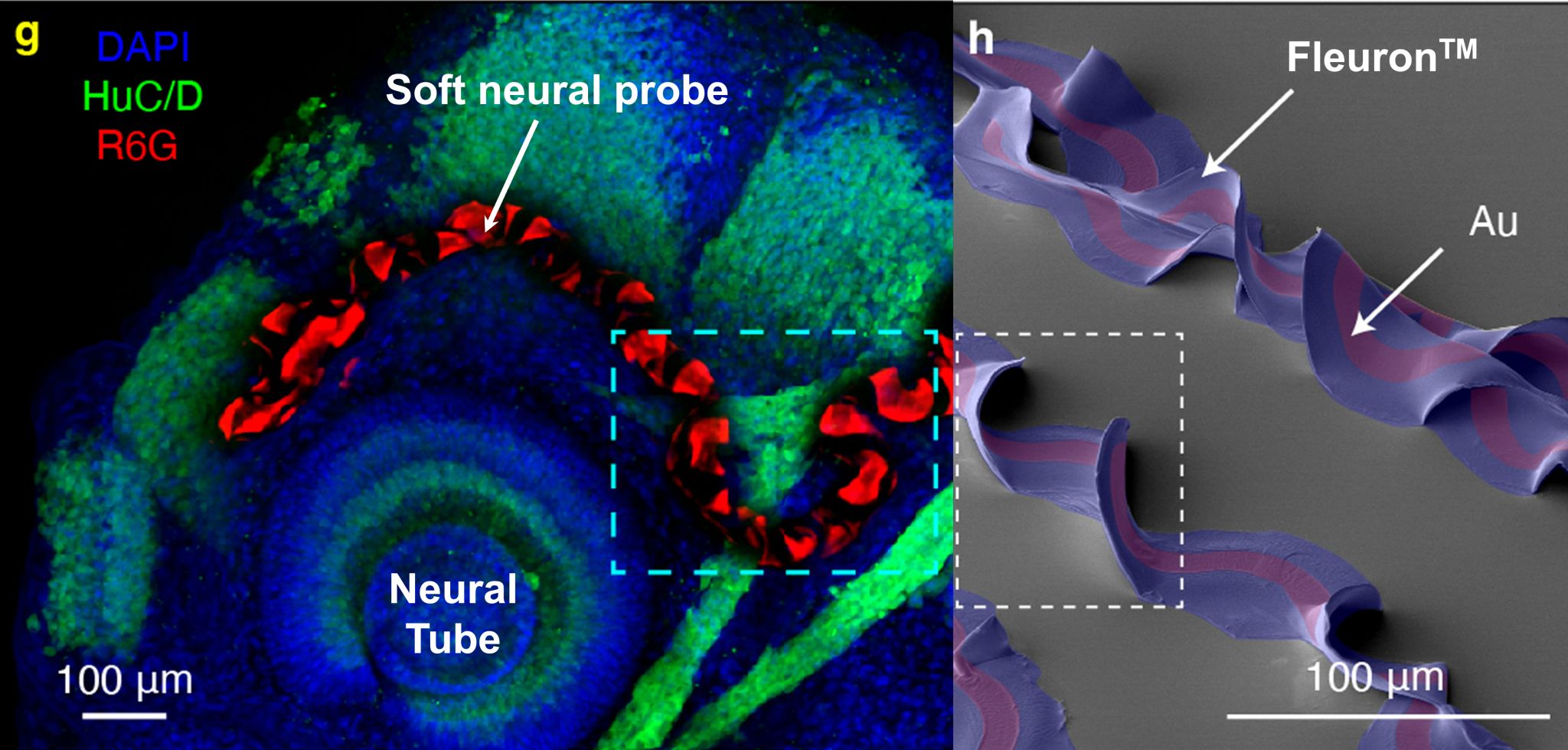
As the brain is an organ consisting of soft, moving, and biologically reactive tissue, materials are key to developing long-lasting implants. Conventional implant electronics have been rigid, creating a mismatch that can contribute to tissue response, scar formation, micromotion damage, signal attenuation, and long-term instability. Axoft’s thesis is that Fleuron can reduce that mismatch. Axoft states that Fleuron is up to 10,000 times softer than polyimide used in existing BCIs, with 8x more region access, 32x more sensors or stimulators per thread, and more than 60% less signal attenuation.
Fleuron is a family of soft perfluorinated elastomers used inside Axoft’s neural probes. It acts as the insulating and protective layer around thin electrode arrays, replacing stiffer materials often used in neural implants. This lets Axoft build probes that are both dense and flexible, rather than trading one for the other. In the underlying Nature Nanotechnology work, the material supported multilayer electrode structures and remained stable in physiological conditions. Axoft has moved that approach into 128-channel human probes designed to access cortical and subcortical tissue, with a path toward 1,024 sensors on a single lead.
Axoft is not alone in chasing better implant materials. INBRAIN Neuroelectronics represents a graphene route, raising $50 million in 2024 to advance its graphene-based BCI-Tx platform and recently completing enrollment in its first-in-human graphene cortical interface study. Neurosoft Bioelectronics is moving through a softer and stretchable cortical interface route. Its SOFT ECoG platform is designed for intraoperative use or implantation for up to 30 days, while its 64-channel interface tested at UMC Utrecht used material described as 1,000 to 100,000 times softer than conventional electrodes.
Even large BCI players are moving toward more biologically integrated ways of interfacing with the brain. Science is developing a biohybrid implant that uses living neurons as an intermediary between device and tissue, with embedded neurons, microLEDs, and electrodes forming a biological bridge with the surrounding brain. The approach is still highly experimental, but it shows how far the interface-layer question is stretching. The field is moving from softer polymers and graphene, to stretchable cortical arrays, to living cellular interfaces designed to make electronic implants behave less like foreign bodies.
[Image credit: Axoft]